Plant Physiology 160: 1551-1566 (2012)
Distinct cell wall architectures in seed endosperms in representatives of the Brassicaceae and Solanaceae [W][OA]
Centre for Plant Sciences, Faculty of Biological Sciences, University of Leeds, Leeds LS2 9JT, UK (KL, PK)
Wageningen Seed Lab, Laboratory of Plant Physiology, Wageningen University, Droevendaalsesteeg 1, 6708 PB, Wageningen, The Netherlands (BD, LB)
Department of Molecular Plant Physiology, Utrecht University, 3584 CH Utrecht, The Netherlands (BD, LB)
University of Freiburg, Faculty of Biology, Institute for Biology II, Botany/Plant Physiology, D-79104 Freiburg, Germany (TS*, GLM*)
ARC Centre of Excellence in Plant Cell Walls, School of Botany, University of Melbourne, Parkville, Victoria 3010, Australia (BD, LB)
* Current Address: School of Biological Sciences, Royal Holloway, University of London, Bourne Building 3-30, Egham, Surrey, TW20 0EX, UK
Received July 13, 2012; Accepted September 4, 2012; Published September 6, 2012.
DOI:10.1104/pp.112.203661
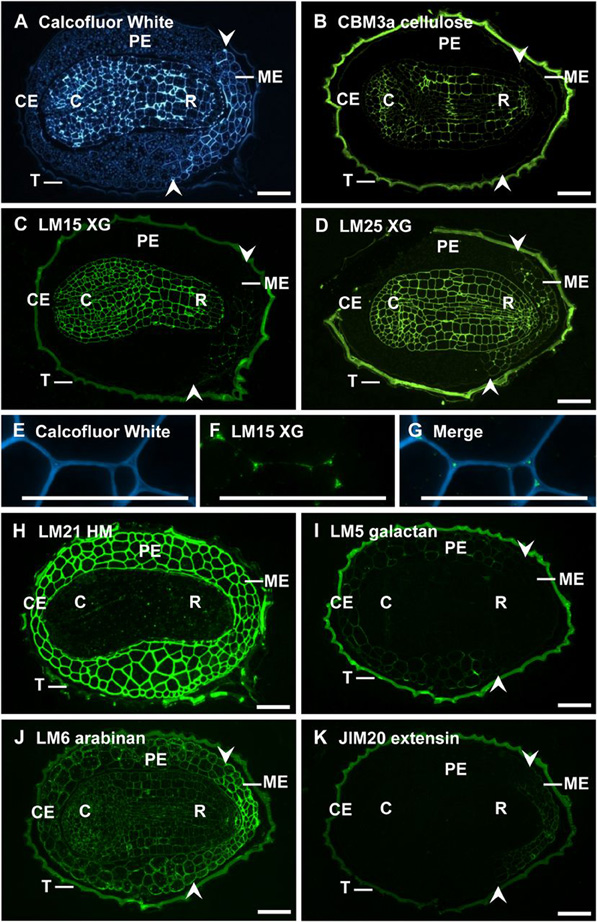
Figure 5. In situ localization of cell wall epitopes in medial longitudinal sections of 3-h-imbibed tobacco seeds.
A, Calcofluor White labeling showing cotyledons (C), radicle (R), testa (T), ME, CE, and PE.
B, CBM3a cellulose labeled the embryo alone.
C and D, LM15 XG and LM25 XG epitopes were uniformly distributed in embryo cell walls and restricted to the ME in the endosperm.
E to G, LM15 XG binds to intercellular spaces and middle lamellar regions in the ME.
H, LM21 HM binds to abundant heteromannans in the endosperm.
I, LM5 galactan is restricted to the PE and CE.
J, LM6 arabinan epitopes are abundant in embryo and endosperm cell walls.
K, Extensin recognized by JIM20 was restricted to the ME. Bars = 50 mm.
| Article in PDF format (1.5 MB) Supplementary data file (2 MB) |
|
|
|
The Seed Biology Place |
Webdesign Gerhard Leubner 2000 |